You can Download Samacheer Kalvi 6th Science Book Solutions Guide Pdf, Tamilnadu State Board help you to revise the complete Syllabus and score more marks in your examinations.
Tamilnadu Samacheer Kalvi 6th Science Solutions Term 2 Chapter 1 Heat
Samacheer Kalvi 6th Science Heat Textual Evaluation
I. Choose the appropriate answer:
Question 1.
When an object is heated, the molecules that make up the object
(a) begin to move faster
(b) lose energy
(c) become heavier
(d) become lighter
Answer:
(a) begin to move faster
Question 2.
The unit of heat is
(a) newton
(b) joule
(c) volt
(d) Celsius
Answer:
(b) Joule
Question 3.
One litre of water at 30°C is mixed with one litre of water at 50°C. The temperature of the mixture will be
(a) 80°C
(b) More than 50°C but less than 80°C
(c) 20°C
(d) around 40°C
Answer:
(d) around 40°C
Question 4.
An iron ball at 50°C is dropped in a mug containing water at 50°C. The heat will
(a) flow from iron ball to water.
(b) not flow from iron ball to water or from water to iron ball.
(c) flow from water to iron ball.
(d) increase the temperature of both.
Answer:
(b) not flow from iron ball to water or from water to iron ball
![]()
II. Fill up the blanks:
- Heat flows from a ______ body to a ______ body.
- The hotness of the object is determined by its ______
- The SI unit of temperature is ______
- Solids on heating and ______ on cooling.
- Two bodies are said to be in the state of thermal ______ if there is no transfer of heat taking place.
Answers:
- higher temperature, lower temperature
- temperature
- kelvin
- expand, contract
- equilibrium
![]()
III. True or False. If False, give the correct statement:
Question 1.
Heat is a kind of energy that flows from a hot body to a cold body.
Answer:
True
Question 2.
Steam is formed when heat is released from water.
Answer:
False. Ice is formed when heat is released from water.
Question 3.
Thermal expansion is always a nuisance.
Answer:
False. Thermal expansion is always beneficial.
Question 4.
Borosilicate glass do not expand much on being heated.
Answer:
True
Question 5.
The unit of heat and temperature are the same.
Answer:
False. The unit of heat and temperature are different
![]()
IV. Give reasons for the following:
Question 1.
An ordinary glass bottle cracks when boiling water is poured into it, but a borosilicate glass bottle does not.
Answer:
The borosilicate glass is pyrex glass. They do not expand much on being heated and therefore they do not crack.
Question 2.
The electric wire which sag in summer become straight in winter.
Answer:
In summer the electric wire is expanded by high temperature. So it sags in summer. In winter die electric wire is contracted by low temperature. Hence it becomes straight in winter.
Question 3.
|Rivet is heated before fixing in the hole to join two metal plates.
Answer:
In order to make the other end of the rivet to form a new “rivet-head” by hammering, the rivet is heated. It becomes malleable when the rivet is in red-hot condition.
![]()
V. Match the following:
| 1. Heat | 0°C |
| 2. Temperature | 100°C |
| 3. Thermal Equilibrium | kelvin |
| 4. Ice cube | No heat flow |
| 5. Boiling water | joule |
Answer:
|
1. Heat |
joule |
| 2. Temperature | kelvin |
| 3. Thermal,Equilibrium | No heat flow |
| 4. Ice cube | 0°C |
| 5. Boiling water | 100°C |
![]()
VI. Analogy:
Question 1.
Heat: Joule :: Temperature : _______
Answer:
kelvin
Question 2.
Ice cube : 0°C :: Boiling water : _______
Answer:
100°C
Question 3.
Total Kinetic Energy of molecules: Heat:: Average Kinetic Energy: _______
Answer:
Temperature
![]()
VII. Give Very Short Answer:
Question 1.
Make a list of electrical equipment at home from which we get heat.
Answer:
The water heater, iron box, electric cattle, etc.,
Question 2.
What is the temperature?
Answer:
- The measurement of the warmness or coldness of a substance is known as temperature.
- SI unit is Kelvin.
- Other units: Celsius, Fahrenheit
Question 3.
What is thermal expansion?
Answer:
The expansion of a substance on heating is called the thermal expansion of that substance.
Question 4.
What do you understand by thermal equilibrium?
Answer:
Thermal equilibrium exists when two objects in thermal contact no longer affect each other’s temperature (or) Both the objects are in same temperature they are in thermal equilibrium.
![]()
VIII. Give Short Answer:
Question 1.
What difference do you think heating the solid will make in their molecules?
Answer:
Heat expands solids. The molecules in the solid move faster, spread apart and occupy more space.
Question 2.
Distinguish between heat and temperature.
Answer:
| S.No, | Heat | Temperature |
|
1. |
Heat not only depends on the temperature of the substance but also depends on how many molecules are there in the object. | Temperature is related to how fast the atoms or molecules move or vibrate within the substance |
| 2. | Heat measures the total Kinetic Energy of the molecules in the substance. | Temperature measures the average kinetic energy of molecules. |
| 3. | SI Unit: Joule | SI Unit: Kelvin |
| 4. | Unit: Joules, Calories | Unit: Fahrenheit, Celsius, Kelvin |
| 5. | It has the ability to do work | It can be used to measure the degree of heat |
![]()
IX. Answer in detail:
Question 1.
Explain thermal expansion with suitable examples.
Answer:
The expansion of a substance on heating is called, the thermal expansion of those substances.
- Gaps are left in between rails while laying on a railway track. The reason is during summer thermal expansion takes place.
- Gaps are left in between two joints of a concrete bridge because of thermal expansion.
- Electric wires between electric posts contract on cold days and sag in summer.
- By fitting the iron rim on the wooden wheel they will heat the iron rim expansion takes place. They will fit with the wheel when the rim is cold, it will not slip.
- Riveting: To join two steel plates together, a hot rivet is driven through the hole of the plates.
One end of the rivet is hammered to form new heat when cooled the rivet will contract and hold tightly.
![]()
X. Questions based on Higher Order Thinking Skills:
Question 1.
When a window is accidentally left open on a winter night, will you feel uncomfortable because the cold is getting in, or because the heat is escaping from the room?
Answer:
1. Heat moves to cold. i.e. Heat energy flows from higher temperature to lower temperature.
2. Hot air rises upwards.
Thinking about these two rules of nature will help us see how air moves in and around our house and how that relates to cooling or heating efficiency.
In the winter, suppose it is 36 degrees Celsius inside our house and 22 degrees Celsius outside. If we open a window, the heat will escape from the room. Like water, heat constantly seeks equilibrium; heat moves to cold until everything is the same temperature. As the heat is escaping from the room, then the cold air is getting into the house through tiny gaps around doors.
Therefore, we feel uncomfortable because the heat Is escaping from the room, based on the rule that heat energy flows from higher temperature to lower temperature.
Question 2.
Suppose your normal body temperature were lower than what it is. How would the sensation of hot and cold change?
Answer:
If the normal body temperature, (37°C or 98.6°F) is lower than what it is, then it is called hypothermia.
In such a case, the body feels cold sensation. When we are too cold, our blood vessels narrow. This reduces blood flow to our skin to save body heat. We may start to shiver. When the muscles tremble this way, it helps to make more heat.
Hypothermia can be a serious or even deadly. Low body temperature usually happens from being out in cold weather. But it may also be caused by alcohol or drug use, going into shock, or certain disorders such as diabetes or low thyroid.
A low body temperature may occur with an infection. This is most common in newborns, older adults, or people who are frail. A very bad infection may also cause an abnormally low body temperature.
Question 3.
If you heat a circular disk with a hole, what change do you expect in the diameter of the hole? Remember that the effect of heating increases the separation between any pair of particles.
Answer:
Many of us “expect” that the diameter of the hole will decrease because the circular disk is free to expand in all directions as well. Note that expansion of metal due to heating is not like gas spreading out wherever space is available. The correct way to think about it is as follows: Hole size is irrelevant in this case. So we can think that hole is very large.
If we carve a hole so large that its radius is slightly less than that of the circular disk itself, then we will have a ring. It is obvious that the radius of the ring will increase, as atoms have more energy so their oscillation amplitude will increase. As the diameter of the ring increases, the diameter of the hole will also increase.
For example, if you want to insert one hallow pipe inside another hollow pipe of the same diameter, just heat anyone and insert the second one into the heated pipe. This explains the answer in an easy way. So it says that diameter will increase with increasing temperature.
![]()
Samacheer Kalvi 6th Science Heat Intext Activities
Activity 1
Question 1.
Take three bowls. Pour very cold water in the first bowl. (you can also add an ice cubes for cooling). Place lukewarm water in the second. Half fill the third with hot water (-not hot enough to burn!)
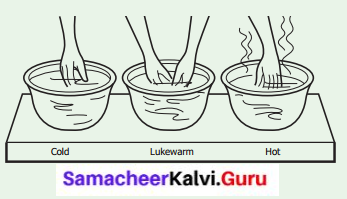
Set them in a row on the table, with the lukewarm water in the center. Place your right hand in the cold water, and your left hand in the hot water. Keep them in for a few minutes. Then take them out, shake off the water, and put both into the middle bowl. How do they feel?
Answer:
I feel that my right hand’s coldness is changed into warm condition because my right hand’s temperature is increased.
I feel that my left hand’s heat is decreased and it is also in warm condition. Here, my left hand’s temperature is decreased.
![]()
Activity 2
Question 1.
The Temperature of Boiling Water.
Take water in a vessel and place the vessel on a stove. Fix the thermometer as shown in figure (Caution: The thermometer should not touch the vessel in which the water is being heated. Otherwise the thermometer will be broken at high temperature.)
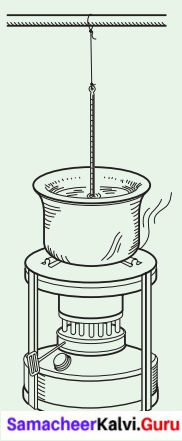
All students have to read the temperature of the water and note the reading on the blackboard. Do you notice that the temperature is rising?
What is the temperature of the water when it is boiling? _______
Answer:
The temperature of the water when boiling is 100°C.
Question 2.
Does the temperature of the boiling water rise further after that?
Answer:
No. the temperature of the boiling water does not rise further after 100°C.
Guess and Write:
(Check your assumption with the help of a thermometer.)
The approximate temperature of the tea when you drink ________
The approximate temperature of cool lemon juice when you drink ________
Answer:
The approximate temperature of the tea when you drink 70°C.
The approximate temperature of cool lemon juice when you drink 20°C.
Question 3.
Is Neela correct?
Beaker A and B have water at 80°C. Then pour the water of A and B to an empty beaker C. Now, What is the A temperature of the water in the beaker C? Neela says it will be 160°C.
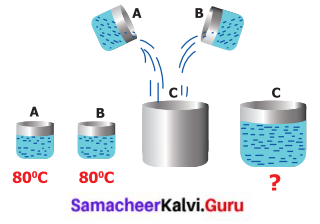
What is your opinion? Does Neela say correctly? Make a guess and verify it experimentally.
Answer:
My opinion is that the temperature will not change. Approximately it will be 80°C only. Neela’s answer is wrong.
Beaker A and Beaker B have water at 80°C. So both beaker has 80°C temperature. Two beakers are in thermal equilibrium. So heat is not transferred to other objects.
![]()
Activity 3
Question 1.
Take one litre of water in a pan, and heat it on a stove. Calculate the time taken to start boiling, (i.e. the time taken to thermometer reading goes up to 100°C). Take five-liter water in another pan and heat it on the same stove. Calculate the time taken by the water to start boiling.
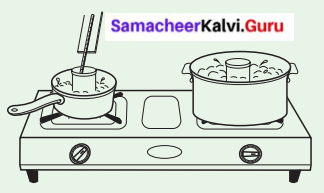
In which pan the water starts to boil earlier?
[ ] One litre water
[ ] Five litre water.
Answer:
[✓] One litre water
Question 2.
Both, however, show a temperature of 100°C at the boiling point. Five-liter water takes more time to boil i.e. more heat is needed to boil the larger amount of water. So, five-liter boiling water has more heat energy than one litre water. Place an open can of lukewarm water in each pan. Observe their temperature to find out which can get hotter.
In which can water show a quick rise in temperature?
[ ] Can in One litre boiled water
[ ] Can in five-liter boiled water.
Answer:
[✓] Can in five-liter boiled water.
Question 3.
You can see that the five-liter water pan will raise the can of water to a higher temperature. Though, both pans of boiling water have a temperature of 100°C the five-liter water can give off more heat energy than one liter water. Because it has more heat energy and gives more energy to the water in the can.
Which has more heat energy in each pair? Put ✓ mark.

The temperature determines the direction of flow of heat.
Question 1.
You are holding a hot cup of coffee, would the Heat energy transfer from

(a) Your body to the coffee, or
(b) The coffee to your body?
Answer:
(b) The coffee to your body
Question 2.
You are standing outside on a summer day. It is 40°C outside (note that normal body temperature is 37°C). Would the Heat energy transfer from.

(a) Your body to the air particles, or
(b) The air particles to your body?
Answer:
(b) The air particles to your body
Question 3.
You are standing outside on a winter day. It is 23°C outside. Would the heat’ energy transfer from:

(a) Your body to the air particles, or
(b) The air particles to your body?
Answer:
(a) Your body to the air particles
![]()
Activity 4
Hammer a nail into a tin can. Ease the nail out. Put it in again to make sure that the hole is large enough for the nail. Then, holding the nail with a pair of pliers, scissors, or forceps, heat the nail over a candle, in hot water, or over the stove. Try to put it into the hole in the can.
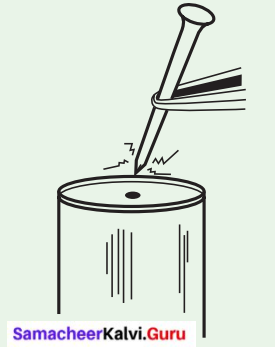
I see that: ________________________________
Answer:
I will see that, now it is hard to put the nail into the hole. Heat expands solids.
The molecules in the solid move faster, spread apart and occupy more space.
Activity 5
Question 1.
Linear Expansion
Take a bulb, dry cell, candle, cycle spoke, coin (or broad-headed nail), and two wooden blocks.
Place one end of the cycle spoke on a wooden block and connect an electric wire to it. Put a stone over the spoke) to hold it firmly in place on the wooden block, as shown in Figure. The spoke should be parallel to the ground. Place the second wooden block under the free end of the spoke. Wrap some electric wire around the coin (or nail) and place it on the block. You may put a stone over the coin to hold it in place.
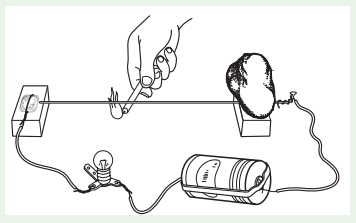
Connect a bulb and dry cell to the free ends of the wires connected to the coin and the spoke and make the circuit shown in the figure.
When the tip of the free end of the spoke touches the coin, the circuit is completed and the bulb lights up. Check to ensure this. If the bulb does not light up, it means the circuit is not complete, so check your connections properly. (Note: We will learn about electric circuit elaborately in electricity lesson.) Now slide a page of your book between the coin and spoke and then slide it out. That way you would get a gap between the coin and spoke equal to the thickness of the sheet of paper,
- Does the bulb light up? If it does not, what could be the reason? ___________________
You saw that the bulb does not light up when the spoke does not touch the coin. Now light the candle and heat the spoke with it. - Did the bulb light up after the spoke was heated for some time?
- If it did, then explain how the spoke touched the coin after it was heated.
- Why does the bulb go off sometime after the candle is taken away from the spoke?
- What happens to the length of the spoke when it is heated or cooled?
Answer:
- The bulbs are not light up, because, the coin and the spoke do not touch it. The circuit is not completed.
- Yes, the bulb is light up, because the spoke is heated the circuit is completed.
- When the spoke is heated, it will be expanded. Here, linear expansion takes place. So it touched the coin.
- After removing the candle, the spoke does not get the heat.
- The spoke is returned to its original state. So it does not touch the coin once again and the bulb goes off.
- The length of the spoke is expanded when the spoke is heated.
![]()
Activity 6
Question 1.
Cubical Expansion
Take a metal ring and metal ball of such size that the ball just passes through the ring.
Heat the ball and check whether it passes through the ring.
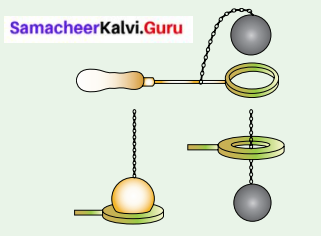
[ ] Passed through
[ ] Not passed through
Answer:
[✓] Not passed through
Question 2.
Now let the ball cool down, and check whether it passes through the ring.
[ ] Passed through
[ ] Not passed through
Answer:
[✓] passed through
Solids expand due to heat and come back to the original state if heat is removed.
Give Reasons for the following
Question 1.
Gaps are left in between rails while laying a railway track.
Answer:

On summer days, the temperature rises and the rails expand. So, in order to allow expansion at the joints of the rails, a small gap is left in between the rails, while laying a railway track. If such a gap is not left at the joints of the rails, the track will get deformed due to thermal expansion. It will cause the derailment of trains.
Question 2.
Gaps are left in between two joints of a concrete bridge.
Answer:

The gaps in the concrete bridges are known as expansion joints, Expansion joints are basically gaps in the bridge that allow the bridge to expand (during summer) and contract (during winter). Without these gaps, the bridge will fall apart.
Question 3.
The photographs below show an expansion joint at the end of a bridge in winter and in summer. Which season is shown in each picture? Explain how do you know?

Answer:
Bridges are made of concrete and steel. These components expand with higher temperature during summer and the gap is minimized in between the joints. So, the summer season is shown in Picture B.
During winter, the components (concrete and steel) contract due to lower temperatures. Therefore the gap is at maximum in between the joints. So, winter season is shown in Picture A.
![]()
Samacheer Kalvi 6th Science Heat Numerical problems
Question 1.
I put a kettle containing 1 litre of cold water on the gas stove, and it takes 5 minutes to reach the boiling point. My friend puts on a small electric kettle, containing 1/2 litre of cold water, and it takes 5 minutes to get up to a boiling point. Which gives more heat in 5 minutes?
a. the gas supply; or
b. the electricity supply? Can you say how many times as much?
Answer:
The gas supply, Two times as much.
Question 2.
One calorie heat energy is needed to raise the temperature of the water from 30°C to 31°C. How much heat energy is needed to raise the temperature of the water from 30°C to 35°C.
Answer:
For 1°C change in temperature, heat energy needed = 1 Calorie
∴ For 5°C change in temperature heat energy needed = 5 Calories.
Samacheer Kalvi 6th Science Heat Additional Questions
I. Choose, the appropriate answer:
Question 1.
We reduce the heat by adding ________ while preparing fruit juice.
(a) sugar
(b) lime
(d) ice cubes
(d) salt
Answer:
(c) ice cubes
Question 2.
The SI unit of temperature is ………..
(a) Calorie
(b) Joule
(c) Celsius
(d) Kelvin
Answer:
(d) Kelvin
Question 3.
Our normal body temperature is _______
(a) 34°C
(b) 36°C
(c) 35°C
(d) 37°C
Answer:
(d) 37°C
Question 4.
Two tumblers of A and B have water at 50°C temperature. If the water from A and B is poured into tumbler C. The temperature of C is ………….
(a) 100° C
(b) 0° C
(c) 50° C
(d) between 0° C to 100° C
Answer:
(c) 50°
Question 5.
______ exists when two objects In thermal contact no longer affect each other’s temperature.
(a) Thermal expansion
(b) Thermal equilibrium
(c) average temperature
(d) coolness
Answer:
(b) Thermal equilibrium
II. Fill in the blanks:
- We feel the heat on our body when the _______ shines.
- _______ energy can be generated by the burning of fuels like coal, wood, charcoal, gasoline etc.
- When _______ flows through a conductor, heat energy is produced.
- _______ is a form of energy.
- _______ determines the direction of the flow of heat.
- The coldest temperature in the world was measured in the _______ continent.
- Temperature measures the _______ kinetic energy of molecules.
Answers:
- Sun
- Heat
- Electric current
- Heat
- Temperature
- Antarctic
- average
![]()
III. True or False. If False, give the correct statement:
Question 1.
The sun gives us light and heat.
Answer:
True
Question 2.
We can absorb heat by rubbing two surfaces of some substances .
Answer:
False. We can generate heat by rubbing two surfaces of some substances.
Question 3.
In the past, people used to rub two wooden pieces together to light fire.
Answer:
False. In the past, people used to rub two stones together to light fire.
Question 4.
When we cool the object the temperature of the object will be increased.
Answer:
False. When we heat the object the temperature of the object will be increased.
Question 5.
Two objects are said to be in thermal contact if they can exchange heat energy.
Answer:
True.
Question 6.
The expansion in volume is called linear expansion.
Answer:
False. The expansion in length is called linear expansion.
![]()
IV. Match the following:
| 1. Source of heat | (a) | heat energy |
| 2. Electric current | (b) | calorie |
| 3. Gasoline | (c) | 37°C |
| 4. Unit of heat | (d) | Electric kettle |
| 5. Human body temperature | (e) | sun |
Answer:
- – e
- – d
- – a
- – b
- – c.
![]()
V. Analogy:
Question 1.
Movement of molecules : Heat.
Heat energy: _______
Answer:
calorie
Question 2.
Expansion is length : Linear expansion.
Expansion in volume : _______
Answer:
cubical expansion
Question 3.
Ordinary glass : Glass tumbler.
Pyrex glass: _______
Answer:
Laboratory glassware.
VI. Give Short Answers:
Question 1.
How can heat energy be generated?
Answer:
Heat energy can be generated by the burning of fuels like wood, Kerosene, Coal, charcoal, gasoline/petrol, oil, etc.
Question 2.
What is heat? Give its units.
Answer:
- Heat is an energy that raises the temperature of a thing by causing the molecules in that thing to move faster.
- Units are Joule, Calorie.
Question 3.
What are the units of heat used?
Answer:
SI unit of heat is Joule. The unit calorie is also used.
Question 4.
Define one calorie.
Answer:
- Total heat is measured by calorie.
- The amount of heat needed to raise one gram of water by one degree centigrade.
Question 5.
Define – Calorie.
Answer:
The amount of heat needed to raise one gram of water by one degree Centigrade.
Question 6.
What is thermal expansion?
Answer:
The expansion of a substance on heating is called the thermal expansion of that substance.
Question 7.
Differentiate linear expansion and cubical expansion.
Answer:
Linear expansion:
- The expansion is in length.
- Ex.: Expansion of railway track rod.
Cubical expansion:
- The expansion is in volume.
- Ex.: Expansion of metal ball.
Question 8.
How solids are expanded on heating?
Answer:
The molecules in the substance move faster when heating, spread apart and occupy more space. So substances expand when heated.
Question 9.
How heat energy is transferred?
Answer:
Heat energy flows from higher temperature to lower temperature.
Question 10.
What type of glassware is used in laboratories? Why?
Answer:
Pyrex glass is used in Laboratory because pyrex glass (Borosilicate glass) do not expand much on being heated and therefore they do not crack.
![]()
VII. Answer in detail.
Question 1.
List and describe the sources of heat.
Answer:
- Sun: It is the primary source of light and it gives us light and heat.
- Combustion: Heat energy can be generated by the burning of fuels like wood, Kerosene, Coal, Charcoal, Petrol, etc.
- Friction: We can generate heat by rubbing two surfaces of some substances.
- In the past, people used to rub two stones together to light fire.
- Electricity: When Electric current flows through a conductor, heat energy is produced. Ex.: Iron box, Electric kettle.